An in-situ Raman Spectroscopic Study on the Interfacial Process of Carbonate-Based Electrolyte on Nanostructured Silver Electrode
Authors
Yu Gu, State Key Laboratory of Physical Chemistry of Solid Surfaces, Innovation Laboratory for Sciences and Technologies of Energy Materials of Fujian Province (IKKEM), College of Chemistry and Chemical Engineering, Xiamen University, Xiamen 361005, China. ;Follow
Yuan-Fei Hu, State Key Laboratory of Physical Chemistry of Solid Surfaces, Innovation Laboratory for Sciences and Technologies of Energy Materials of Fujian Province (IKKEM), College of Chemistry and Chemical Engineering, Xiamen University, Xiamen 361005, China. ;
Wei-Wei Wang, State Key Laboratory of Physical Chemistry of Solid Surfaces, Innovation Laboratory for Sciences and Technologies of Energy Materials of Fujian Province (IKKEM), College of Chemistry and Chemical Engineering, Xiamen University, Xiamen 361005, China. ;
En-Ming You, State Key Laboratory of Physical Chemistry of Solid Surfaces, Innovation Laboratory for Sciences and Technologies of Energy Materials of Fujian Province (IKKEM), College of Chemistry and Chemical Engineering, Xiamen University, Xiamen 361005, China. ;
Shuai Tang, College of Chemistry, Zhengzhou University, Zhengzhou 450001, China
Jian-Jia Su, State Key Laboratory of Physical Chemistry of Solid Surfaces, Innovation Laboratory for Sciences and Technologies of Energy Materials of Fujian Province (IKKEM), College of Chemistry and Chemical Engineering, Xiamen University, Xiamen 361005, China. ;
Jun-Yin Jun-Yin, State Key Laboratory of Physical Chemistry of Solid Surfaces, Innovation Laboratory for Sciences and Technologies of Energy Materials of Fujian Province (IKKEM), College of Chemistry and Chemical Engineering, Xiamen University, Xiamen 361005, China.
Jia-Wei Yan, State Key Laboratory of Physical Chemistry of Solid Surfaces, Innovation Laboratory for Sciences and Technologies of Energy Materials of Fujian Province (IKKEM), College of Chemistry and Chemical Engineering, Xiamen University, Xiamen 361005, China.
Zhong-Qun Tian, State Key Laboratory of Physical Chemistry of Solid Surfaces, Innovation Laboratory for Sciences and Technologies of Energy Materials of Fujian Province (IKKEM), College of Chemistry and Chemical Engineering, Xiamen University, Xiamen 361005, China. ;
Bing-Wei Mao, State Key Laboratory of Physical Chemistry of Solid Surfaces, Innovation Laboratory for Sciences and Technologies of Energy Materials of Fujian Province (IKKEM), College of Chemistry and Chemical Engineering, Xiamen University, Xiamen 361005, China. ;Follow
Corresponding Author(s)
Yu Gu(ygu@xmu.edu.cn);
Bing-Wei Mao(bwmao@xmu.edu.cn)
Abstract
The solid-electrolyte interphase (SEI) plays a key role in anodes for rechargeable lithium-based battery technologies. However, a thorough understanding of the mechanisms of SEI formation and evolution remains a major challenge, hindering the fast development and wide applications of Li-based batteries. Here, we devise a borrowing surface-enhanced Raman scattering (SERS) activity strategy from size optimized Ag nanosubstrate to in-situ monitor the SEI formation and evolution and its structure and chemistry in an ethylene carbonate-based electrolyte. To ensure a reliable in-situ SERS investigations, we design a strict air-tight Raman cell with three-electrode configuration. Based on the potential-dependent spectroscopic information, we reveal that the SEI formed in EC-based electrolyte presents a double-layer structure that consists of a thin inorganic inner layer and an organic-rich outer layer. We also exclude that LEMC is the major component of EC reduction rather than LEDC and the critical role of metallic Li in the formation of stable SEI is preliminary explored. Nevertheless, the identification of the SEI compositions can be easily done only before the Li deposition on Ag surface. After the formation of Li–Ag alloys, the subsequent evolution of SEI could not be detected due to the dielectric constant change of Ag after the lithiation. Our work provides a real-time spectroscopic method for investigation of interfacial processes of anodes, which is beneficial to the understanding of SEI formation and evolution and thus provides guidance for development of rationally designed SEI in Li-based batteries.
Graphical Abstract
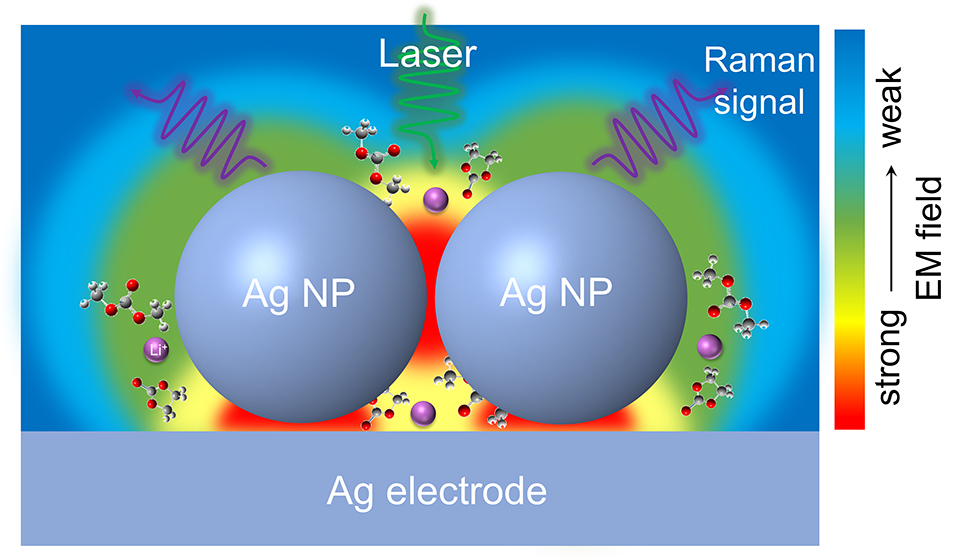
Keywords
solid-electrolyte interphase, in-situ SERS, anodes, lithium-based batteries
DOI
10.13208/j.electrochem.2301261
Recommended Citation
Yu Gu, Yuan-Fei Hu, Wei-Wei Wang, En-Ming You, Shuai Tang, Jian-Jia Su, Jun-Yi, Jia-Wei Yan, Zhong-Qun Tian, Bing-Wei Mao. An in-situ Raman Spectroscopic Study on the Interfacial Process of Carbonate-Based Electrolyte on Nanostructured Silver Electrode[J]. Journal of Electrochemistry, doi: 10.13208/j.electrochem.2301261.

